PBIS — Pharmaceutical Polybag Inspection System


Pinholes, seal spread, and particle contamination invisible to manual inspection cause moisture ingress and API degradation during storage.
Defective polybags discovered during stability testing result in costly batch rejections and regulatory holds for pharmaceutical manufacturers.
21 CFR Part 11, GDP, and ICH Q7 require 100% inspection of pharmaceutical packaging. Manual methods are unreliable and not audit-proof.


Pharmaceutical polybags are large flexible packaging made from LDPE, HDPE, or aluminium-foil laminates. They hold APIs (Active Pharmaceutical Ingredients), finished tablets, capsules, and powder intermediates before drumming and further packaging. Polybag integrity is critical for product stability and shelf-life.
A single defect in a polybag—a pinhole, weak seal, or particle—can compromise the entire batch during stability testing. Moisture ingress degrades active ingredients. Particle contamination risks customer safety. Regulatory bodies expect 100% inspection evidence.
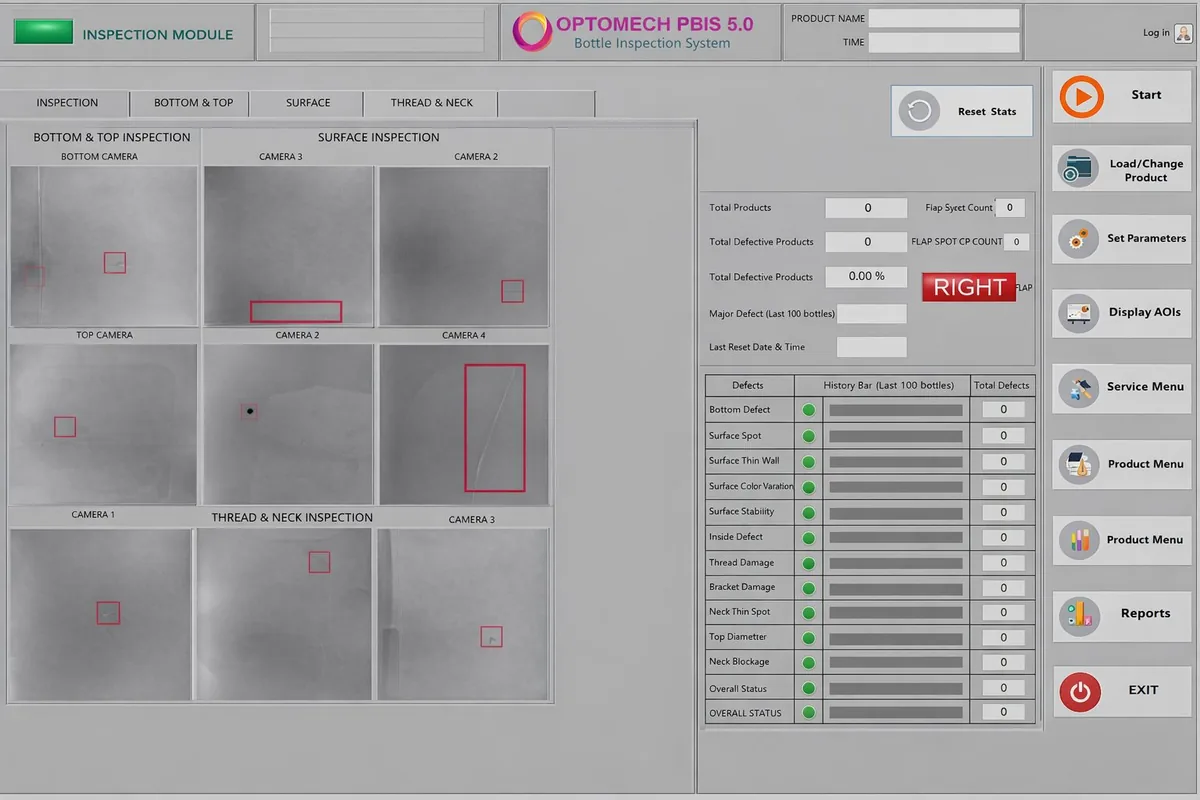
PBIS uses 9 high-resolution cameras positioned at multiple angles around each polybag. AI-powered deep learning algorithms analyze images in real-time, detecting all 6 critical defect types with >99% accuracy. Pass/Fail sorting and complete traceability are automatic.
Total Field of View: 1385 mm × 1070 mm
PBIS covers an inspection area of 1385 mm × 1070 mm — meaning any polybag up to this size is fully inspected in a single pass, in one shot. All defects are detected across the entire surface without multiple scans or repositioning.
Microscopic perforations or larger punctures allowing moisture and oxygen ingress, compromising API stability.
Inadequate heat sealing or seal defects that fail to provide hermetic closure and allow contamination.
Foreign particles (dust, fibres, fragments) embedded in or on the polybag surface during manufacturing.
Discoloration, delamination, or oxidation marks larger than 0.6mm indicating material degradation or contamination.
Dark spots or burn marks on the polybag surface exceeding 0.4mm, caused by heat or chemical degradation.
Tears or cuts in the polybag material that compromise structural integrity and barrier properties.

Holes & Punctures

Seal Spread

Particle Contamination

White Spots >0.6mm

Black Spots >0.4mm

Cuts in Material
Get complete technical specifications, compliance documentation, ROI analysis, and integration guidelines for the Polybag Inspection System.
Polybags are fed into the PBIS system via conveyor belt. Position sensors ensure correct alignment for multi-angle imaging.
9 high-resolution cameras capture detailed images of the polybag surface, seams, and entire perimeter simultaneously.
AI algorithms analyze all images in real-time, identifying all 6 defect types with sub-millimeter accuracy and minimal false rejections.
Defective bags are automatically sorted and rejected. Complete inspection reports, timestamps, and defect images are logged for traceability.
PBIS uses 9 strategically positioned high-resolution cameras to capture polybags from multiple angles simultaneously. This ensures no defect goes undetected, even on curved or embossed surfaces.
Our proprietary AI models are trained on thousands of real pharmaceutical polybag images, both defective and compliant. The system learns to detect not just known defect patterns, but novel defects that emerge in production.
Defect thresholds (hole size, spot dimensions, seal width) are fully configurable by operators. The system learns from your specific material types and quality standards, adapting detection sensitivity in real-time.
Advanced algorithms distinguish between true defects and surface artifacts (dust, lighting reflections). This dramatically reduces false rejections that waste product and reduce throughput.
9 synchronized cameras ensure 360 coverage with no blind spots.
Results in <100ms, supporting 3,000 bags/hour throughput.
System learns from production data and improves accuracy over time.
Adjust sensitivity per material type, batch, or customer requirement.
Identifies new defect patterns not in the training dataset.
21 CFR Part 11 compliant logging of all decisions and images.
PBIS maintains electronic records with digital signatures, audit trails, and data integrity controls meeting FDA requirements for computerized systems.
Good Distribution Practice requirements for 100% packaging inspection are fully satisfied by PBIS's automated, documented inspection process.
ICH Quality Guidelines for Active Pharmaceutical Ingredients require robust quality checks on bulk packaging—PBIS delivers certified compliance.
Every inspected bag is logged with timestamp, defect data, images, and pass/fail status. Full traceability for recalls and audits.
9 industrial-grade cameras with 12MP+ sensors for sub-millimeter defect detection across all polybag materials.
GPU-accelerated deep learning inference processes images in <100ms, achieving 3,000 bags/hour throughput.
Industrial cameras, LED ring lights, vision controllers, and framegrabbers integrated for reliable 24/7 operation.
Data sync to cloud for analytics, reporting, and remote monitoring. Local processing for offline operation and low-latency sorting.
Compatible with most polybag conveyor systems. Custom integration packages available for existing lines.
OPC-UA, REST APIs, and MES integration for seamless manufacturing execution and predictive analytics.
Pharmaceutical bulk API suppliers require 100% polybag inspection for stability testing and regulatory compliance across batches.
Finished tablet, capsule, and powder manufacturers use PBIS for intermediate packaging quality assurance before final drumming.
Food, cosmetic, and consumer goods manufacturers benefit from PBIS's defect detection for polybag quality control.
Pesticide and fertilizer manufacturers inspect polybag packaging for moisture and contamination risks during storage and distribution.
Chemical, dye, and industrial material manufacturers rely on PBIS for polybag integrity verification before shipment.
Manufacturers of large Plate Heat Exchanger (PHE) Gaskets use PBIS to inspect rubber and polymer sheets for surface defects, dimensional accuracy, and material integrity before assembly.
Industries manufacturing large metal stamping parts rely on PBIS's wide field-of-view inspection to detect surface defects, burrs, cracks, and dimensional non-conformances in a single shot.
20+ years of pharmaceutical vision inspection. We understand 21 CFR Part 11, GDP, ICH Q7, and real production challenges.
Our deep learning models are trained on real pharmaceutical data. >99% accuracy with minimal false rejections on production lines.
PBIS integrates seamlessly with existing conveyor systems and MES platforms. Installation, training, and support included.
Ready-made 21 CFR Part 11 validation packages, SOPs, and audit templates. We handle the compliance heavy lifting.
24/7 technical support, remote diagnostics, software updates, and on-site training. Your success is our priority.
PBIS typically pays for itself in 18-24 months through reduced batch rejections, lower rework costs, and faster throughput.
PBIS inspects LDPE (Low-Density Polyethylene), HDPE (High-Density Polyethylene), and aluminium-foil laminate polybags used in pharmaceutical bulk drug packaging, API storage, and intermediate product packaging.
PBIS detects holes and defects as small as 0.2mm, white spots >0.6mm, black spots >0.4mm, and particle contamination. Thresholds are fully configurable based on material type and customer requirements.
PBIS inspects up to 3,000 polybags per hour with real-time AI analysis and automated sorting. Actual throughput depends on conveyor speed, polybag size, and integration configuration.
Yes. PBIS is fully compliant with 21 CFR Part 11 (FDA regulations for electronic systems), GDP (Good Distribution Practice), and ICH Q7 (Quality Guidelines). Complete audit trails, digital signatures, and data integrity controls are built-in.
PBIS connects to most standard polybag conveyor systems via mechanical interface and PLC integration. We provide custom adapters, electrical integration, and MES/cloud connectivity. Full commissioning and validation support is included.
Most PBIS installations achieve ROI within 18-24 months through reduced batch rejections, lower rework costs, eliminated recall risks, faster throughput, and reduced labor. Contact our sales team for a detailed ROI analysis for your operation.
PBIS detects 6 primary defect types: holes, seal spread, particle contamination, white spots, black spots, and cuts. Additionally, the deep learning algorithm can identify novel or unusual defects not in the training dataset, providing comprehensive quality control beyond preset thresholds.
Optomech provides on-site operator and maintenance training, complete documentation, SOP templates, 21 CFR Part 11 validation packages, and 24/7 technical support. Software updates, remote diagnostics, and annual preventive maintenance are available as service packages.
Yes. PBIS supports cloud integration for real-time dashboards, historical trend analysis, batch reporting, and predictive maintenance. All data is encrypted and compliant with data protection regulations. Local operation is also available for offline or secure environments.
Schedule a live demo of PBIS with your production samples. See real-time AI detection, 3,000 bags/hour throughput, and 21 CFR Part 11 compliance in action.