FOSIS — Flip-Off Seal Inspection System


Defect types detected across 3 zones for comprehensive quality assurance
Automatic inspection with zero manual intervention required
Full FDA compliance for pharmaceutical manufacturing standards



A flip-off seal is a tamper-evident closure mechanism used in pharmaceutical packaging. It consists of an aluminium shell that covers and protects the product, with a button designed to be flipped off to access the contents. These seals are critical for maintaining product integrity, sterility, and compliance.
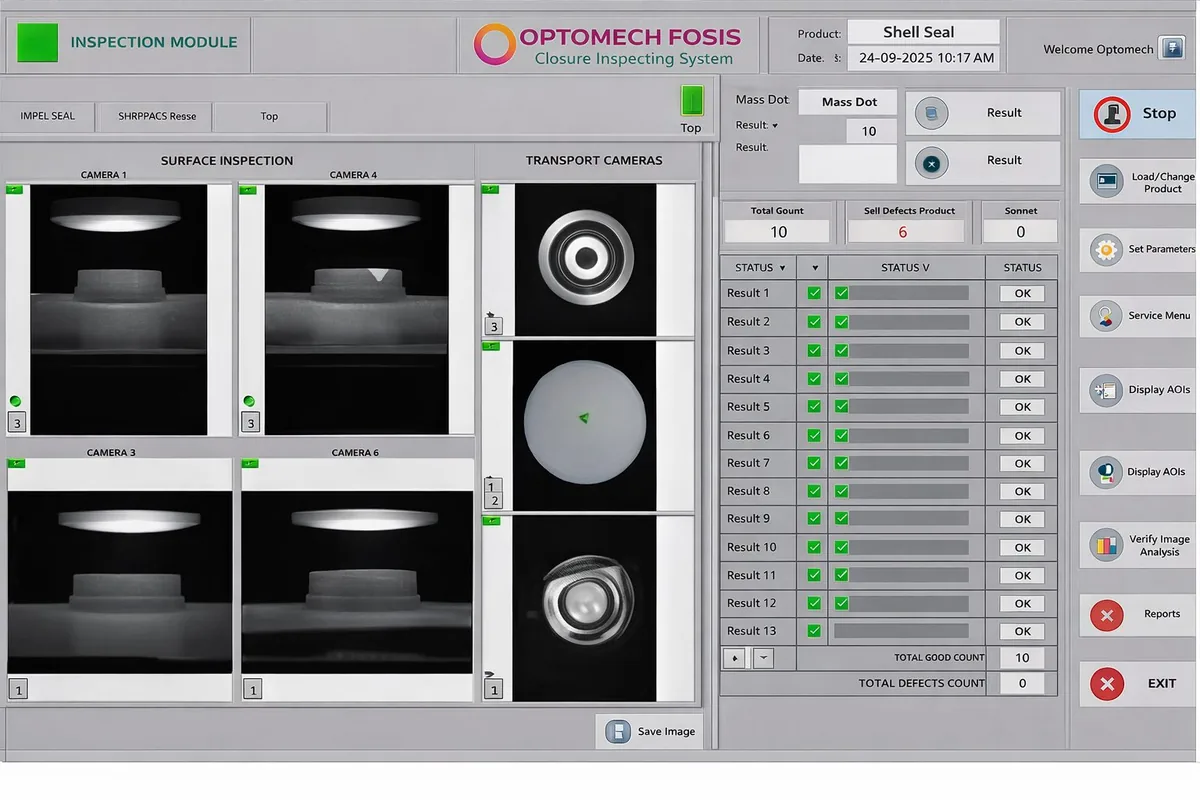
FOSIS uses advanced machine vision and AI deep learning to inspect flip-off seals at high speed. The system automatically detects defects across three zones: the outer aluminium shell, inner surface, and PP button. Defective items are instantly ejected using pneumatic ejection, ensuring 100% quality control.








Get detailed specifications, technical data, and application information
Vibratory SS304 bowl feeder automatically orients and conveys seals to the inspection line
Advanced vision system captures high-resolution images of all three zones simultaneously
Deep learning algorithms analyze images and identify 18+ defect types in milliseconds
Defective items ejected pneumatically; data logged with images for complete traceability
FDA compliant electronic records and signatures for pharmaceutical manufacturing
Operator, Supervisor, and Administrator roles with secure login and audit trails
Meets international GMP standards for pharmaceutical manufacturing quality
Last 100 defects stored with images; full data export for batch validation
High-resolution multi-camera system capturing all seal zones with precision optics
Neural networks trained on thousands of defect examples for accurate detection
Optimized LED lighting to highlight defects across all three inspection zones
Intuitive operator interface showing live inspection, statistics, and defect images
High-speed pneumatic system for instant rejection of defective seals
Secure cloud connectivity for software updates and remote diagnostics
20+ years of pharmaceutical machine vision experience and proven track record
21 CFR Part 11 certified systems with comprehensive validation documentation
24/7 technical support, spare parts availability, and on-site service worldwide
Deep learning algorithms continuously improve defect detection accuracy
Flexible configurations for different seal sizes and defect requirements
Fast payback period with reduced waste, improved yield, and faster cycle times
See the system in action and learn how it can improve your pharmaceutical manufacturing process