Frequently Asked Questions
What is CANIS Canister Inspection System?
+
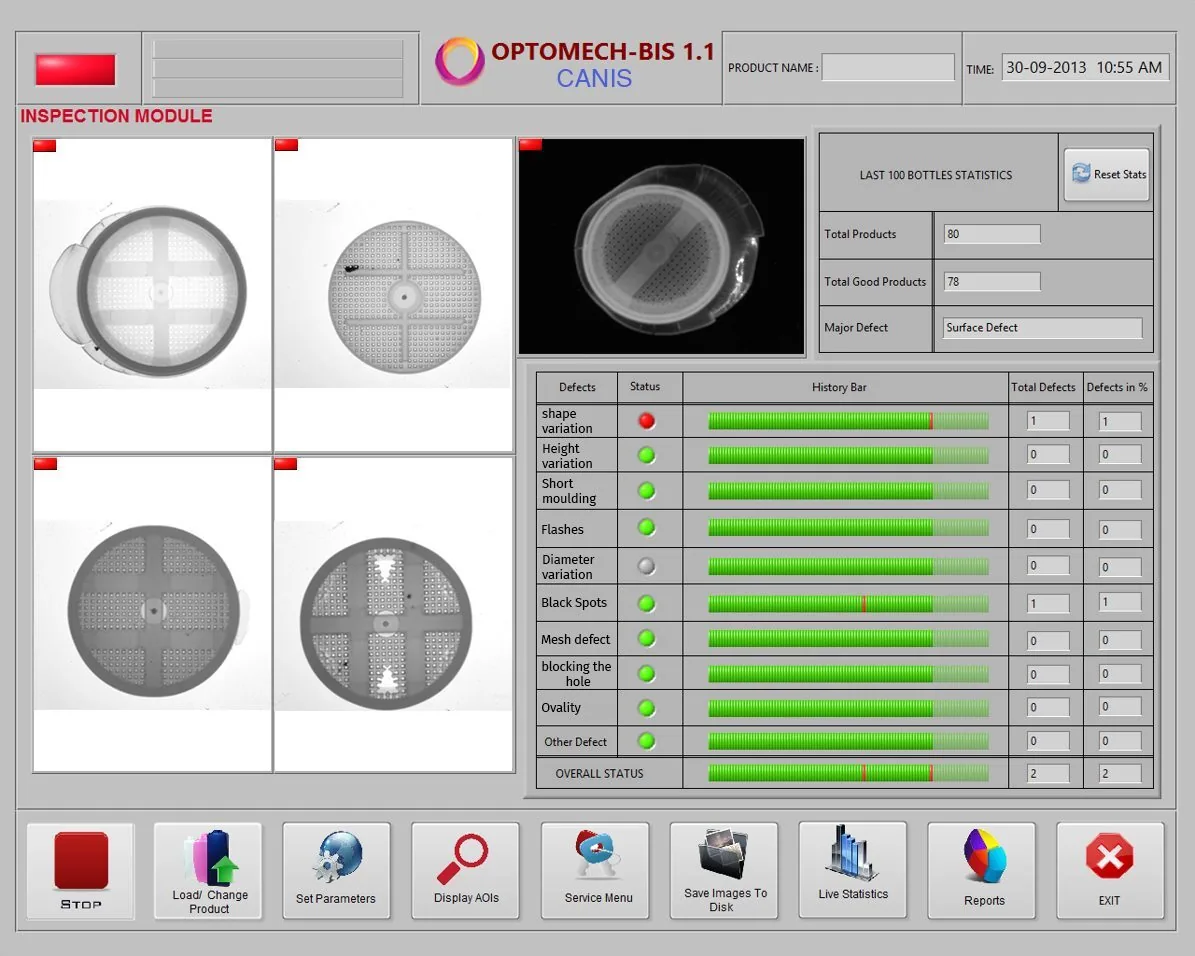
CANIS is an AI-powered vision inspection system that detects defects in silica desiccant canisters used in pharmaceutical bottles. It inspects 14,000 canisters per hour and detects 11 types of defects including the critical mesh breakage, ensuring 100% GMP compliance.
What is mesh breakage and why is it critical?
+
Mesh breakage occurs when the protective mesh layer of the desiccant canister breaks, allowing silica beads to escape and contaminate the drug product. This is a GMP critical defect that triggers immediate batch quarantine, recalls, and regulatory action.
What defects does CANIS detect?
+
CANIS detects 11 types of defects across 2 categories: Structural Defects (mesh breakage, flashes, short moulding, ovality, improper cap, reverse cap) and Label/Surface Defects (no label, label width deviation, label shifts, label fold, double label, black spots).
What is the inspection speed of CANIS?
+
CANIS inspects 14,000 canisters per hour with 100% inspection coverage and >99.8% defect detection accuracy, enabling high-speed pharmaceutical production lines without bottlenecks.
Is CANIS compliant with pharmaceutical regulations?
+
Yes, CANIS is fully compliant with Schedule M (Revised 2025), WHO-GMP Annex 2, and ISO standards. It maintains complete audit trails, batch traceability, and defect documentation required for regulatory compliance.
What types of canisters does CANIS inspect?
+
CANIS inspects silica desiccant canisters of various sizes, shapes, and materials used in pharmaceutical oral solid dosage forms (tablets and capsules). The system is configurable for different canister geometries.
Can CANIS provide defect traceability?
+
Yes, CANIS provides complete traceability with batch-to-canister mapping, serial numbers, timestamps, defect images, operator IDs, and digital reports. All data is stored for regulatory audits and GMP compliance documentation.
What industries use CANIS?
+
CANIS is used by oral solid dosage pharma manufacturers, desiccant canister manufacturers, API & bulk drug producers, and injectable pharmaceutical companies. It's trusted by leading manufacturers globally.
How does CANIS differ from manual inspection?
+
CANIS provides consistent 24/7 inspection at 14,000 canisters/hour without fatigue. It detects microscopic defects that human inspectors miss, ensures zero bias, and produces complete audit trails for compliance documentation.
What is the typical ROI for CANIS?
+
Most pharma customers see ROI within 12-18 months through reduced recall costs, prevention of batch losses, eliminated manual inspection labor, and increased production throughput at high speeds.