Induction Seal Verification System

- Optomech Induction Seal Verification System uses cutting edge infrared imaging technology to inspect and reject induction sealed containers.
- A good induction seal prevents product leakage, is tamper proof, maintains product quality and fosters consumers’ confidence. Primary packaging sealing integrity is crucial in ensuring product viability and shelf life.
- The days of sampling as the primary means of inspection and analysis for quality control is nearing an end. Manufacturers are looking for 100% inspection of sealed containers, the ISIVS (Induction sealing integrity verification system) does non-destructive 100% inspection by using dynamic thermal imaging to inspect the integrity of induction sealed bottles.
- Inspection is performed through closed bottle caps without damaging or degrading the product and without reducing lining speeds.
OPERATING PRINCIPAL
When the induction sealed container passes through out the ISIVS a highly sensitive thermal camera images the heat profile of the seal. Advanced software algorithms are used to analyze the thermal image for detecting the defects. The defective containers are ejected from the line.
TYPICAL DEFECTS












Typical Defects
- Cut Liner.
- Loose Cap.
- Moon Cut.
- No Liner.
- Over Heating.
- Under Heating.
Learn More
Salient features
- Extremely clear and sharp images using advanced thermal imaging technology.
- Large 17” industrial touch screen user interface.
- 2 way ejection mechanism to ensure no defective product is allowed to pass.
- Ejection bin is provided with level sensors.
- All parts are made with stainless steel.
- Any number of different products can be configured and stored directly by the user.
- Tolerances for the acceptance and rejection criteria can be set and saved in the database.
- Changeover is simple process of just selecting the product from the list of the configured products.
- Remote online support is provided by Optomech, for help in configuring new products.
- Software complies with USFDA code of federal regulations (Title 21 CFR Part 11).
- Multi level password protected user access control is provided.
- Feeding wheel integration is provided.
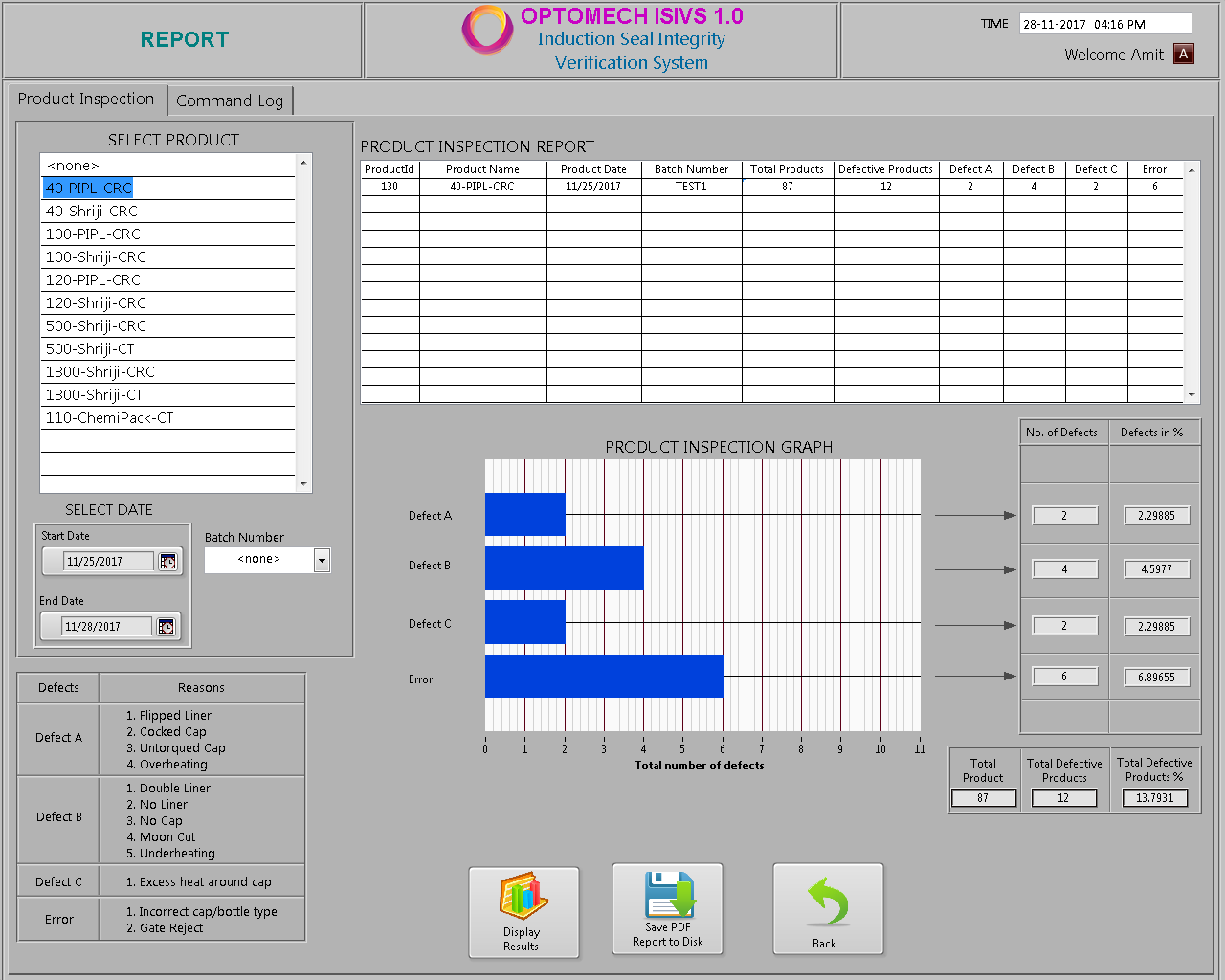
Reporting
- The inspection screen displays graphically the last 100 detected defects to enable you to discover and fix potential problems in real time.
- All inspection data/results is stored in the database.
- Reports enable statistical process control of the induction sealing Process over time.
- User access command log report can be generated.
- Reports can be generated in both PDF and Excel formats.
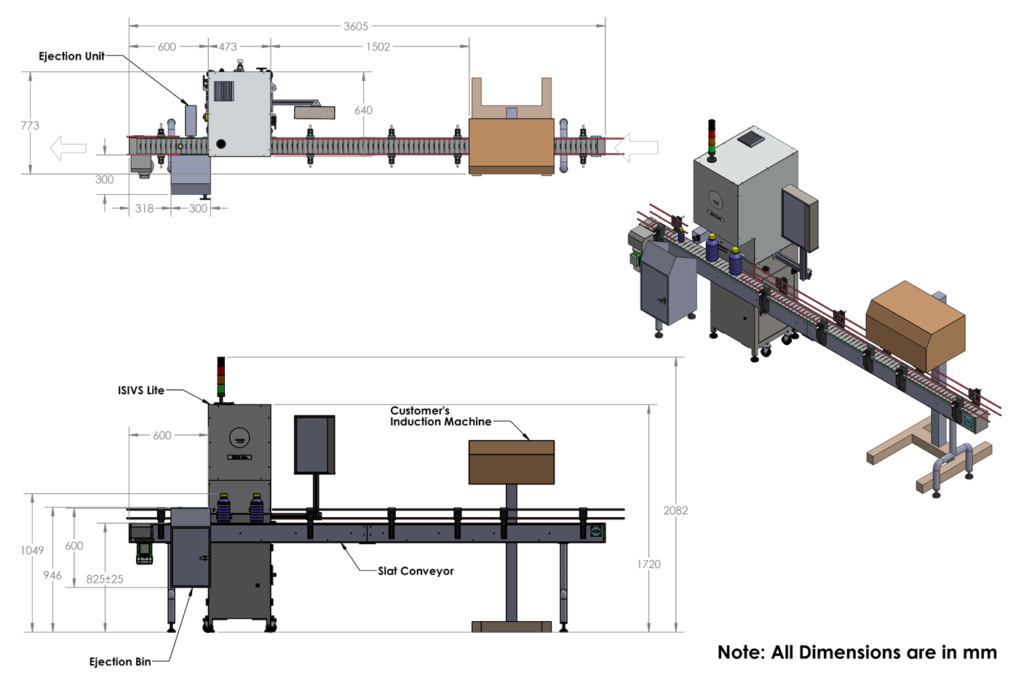
Machine Dimension
Software Screenshot