Eye Dropper bottle inspection System

- Optomech BIS-EDB uses cutting edge imaging technology to inspect and reject defective Eyedropper bottles.
- A good Eye dropper bottle prevents product leakage, is tamper proof, maintains product quality and fosters consumers’ confidence. Primary packaging product quality is crucial in ensuring product viability and shelf life.
- The days of sampling as the primary means of inspection and analysis for quality control is nearing an end. Manufacturers are looking for 100% inspection of containers, the BIS-EDB ( Eyedropper bottle inspection system) does 100% inspection of Eyedropper bottles using high resolution machine vision cameras to inspect the quality of eyedropper bottles.
- Inspection is performed at very high speeds and defective products are ejected off the line.
OPERATING PRINCIPAL
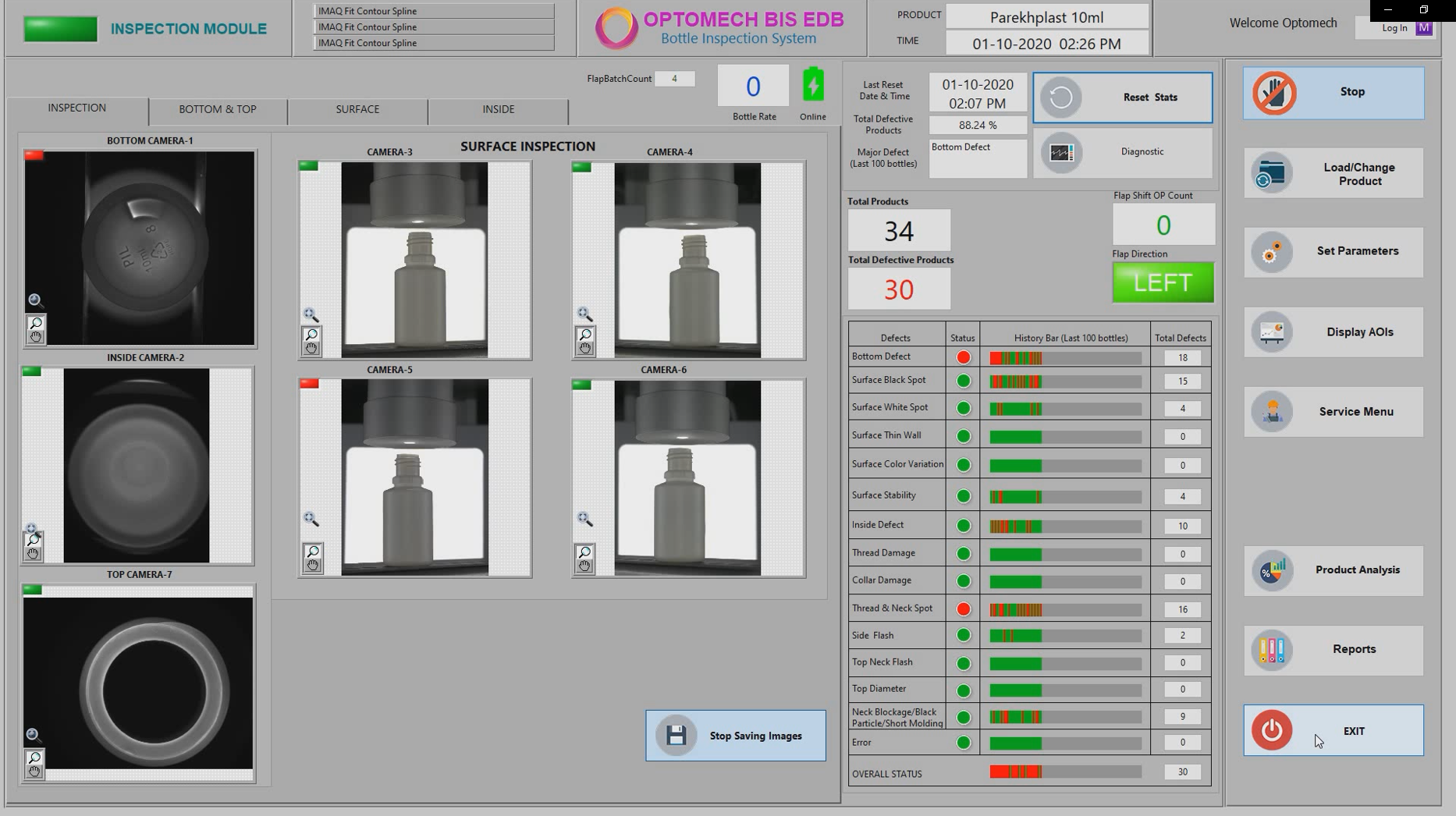
The Eye dropper bottles pass through the BIS-EDB’s multiple highly resolution machine vision cameras, images are captured by the high resolution machine vision cameras. Advanced software algorithms are used to analyze the images for detecting the defects. The defective bottles are ejected from the line.
TYPICAL DEFECTS






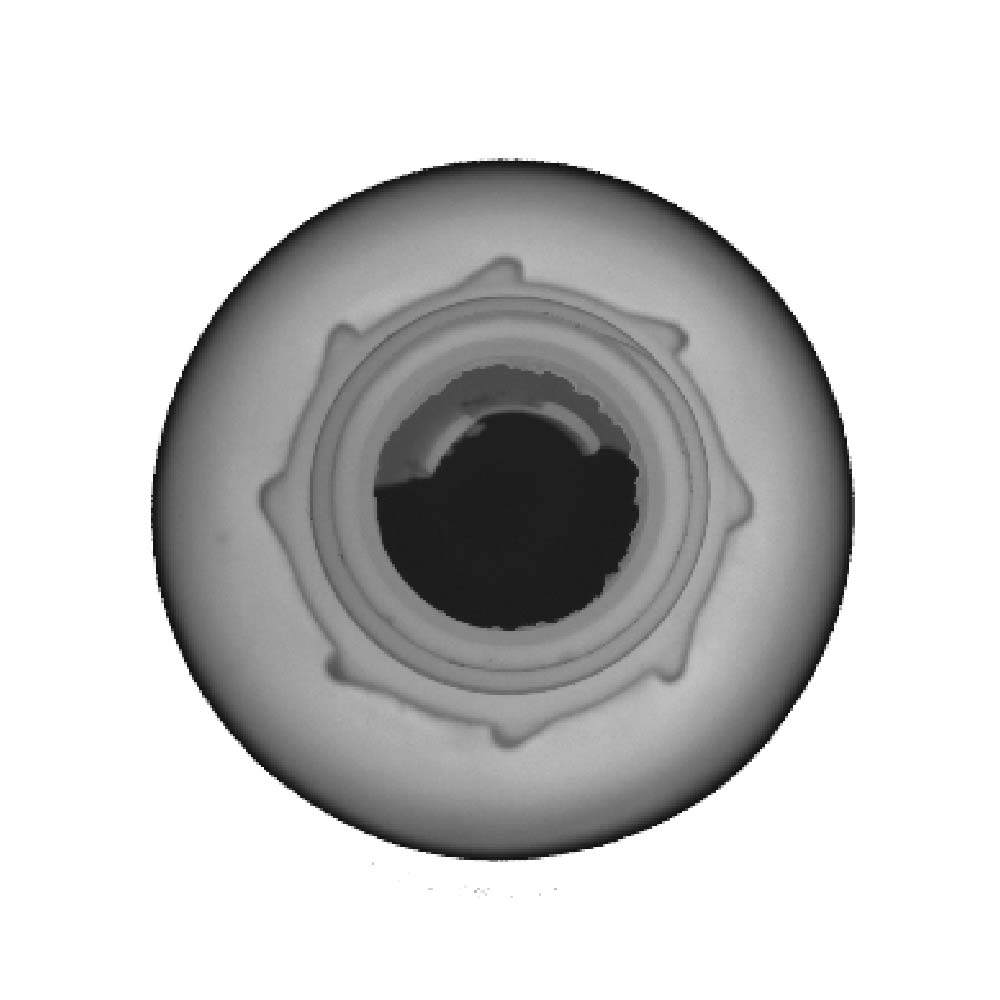














Typical Defects
- Black Spots, material inclusions, burn Spots
- Flashes/Extra material.
- Groove Shear.
- Oil Contamination.
- Holes and thin walls.
- Side weak.
- Incomplete blowing or bottle shape variation.
- Parting line flash.
- Discoloration (Major)
- Double Shot
- Preform
- Shrinkage
- Short fill
- Mixed product separation
- Holes/ bottom burst
- Thin wall/ bottom weak
- Bottom Logo Missing
- Bottom flash
- Bottom bulge
- Ratchet not blow.
- Neck Contamination
Learn More
Salient features
- Extremely clear and sharp images using high speed machine vision cameras.
- Precision optics and customized lighting.
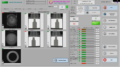
- Large 21” industrial touch screen user interface, displays the image of the seals as they are passing through the system.
- High speed pneumatic ejection.
- Any number of different products can be configured and stored directly by the user.
- Tolerances for the acceptance and rejection criteria can be set and saved in the database.
- Changeover is simple process of just selecting the product from the list of the configured products.
- Remote online support is provided by Optomech, for help in configuring new products.
- Software complies with USFDA code of federal regulations (Title 21 CFR Part 11).
- Multi level password protected user access control is provided.
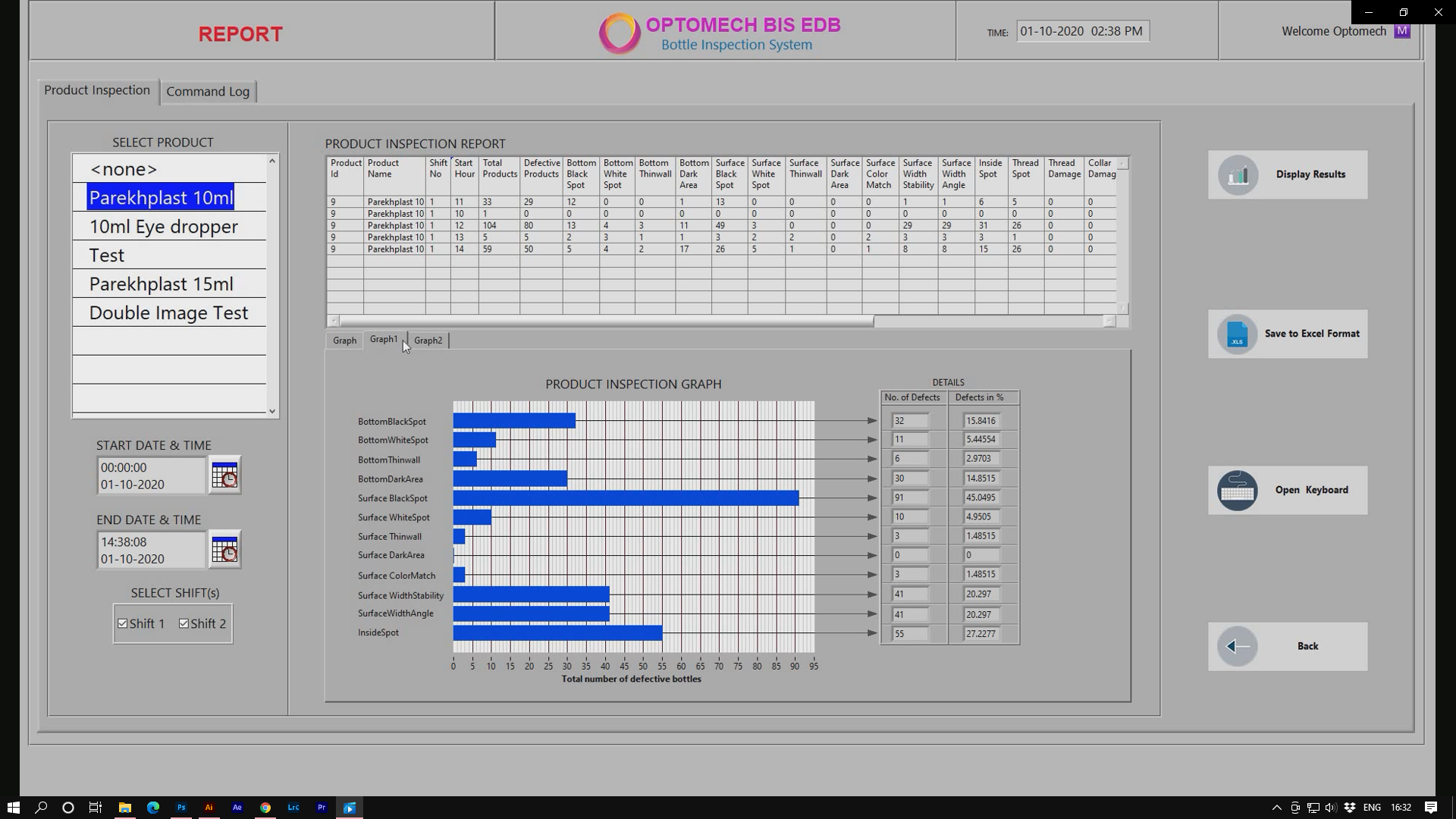
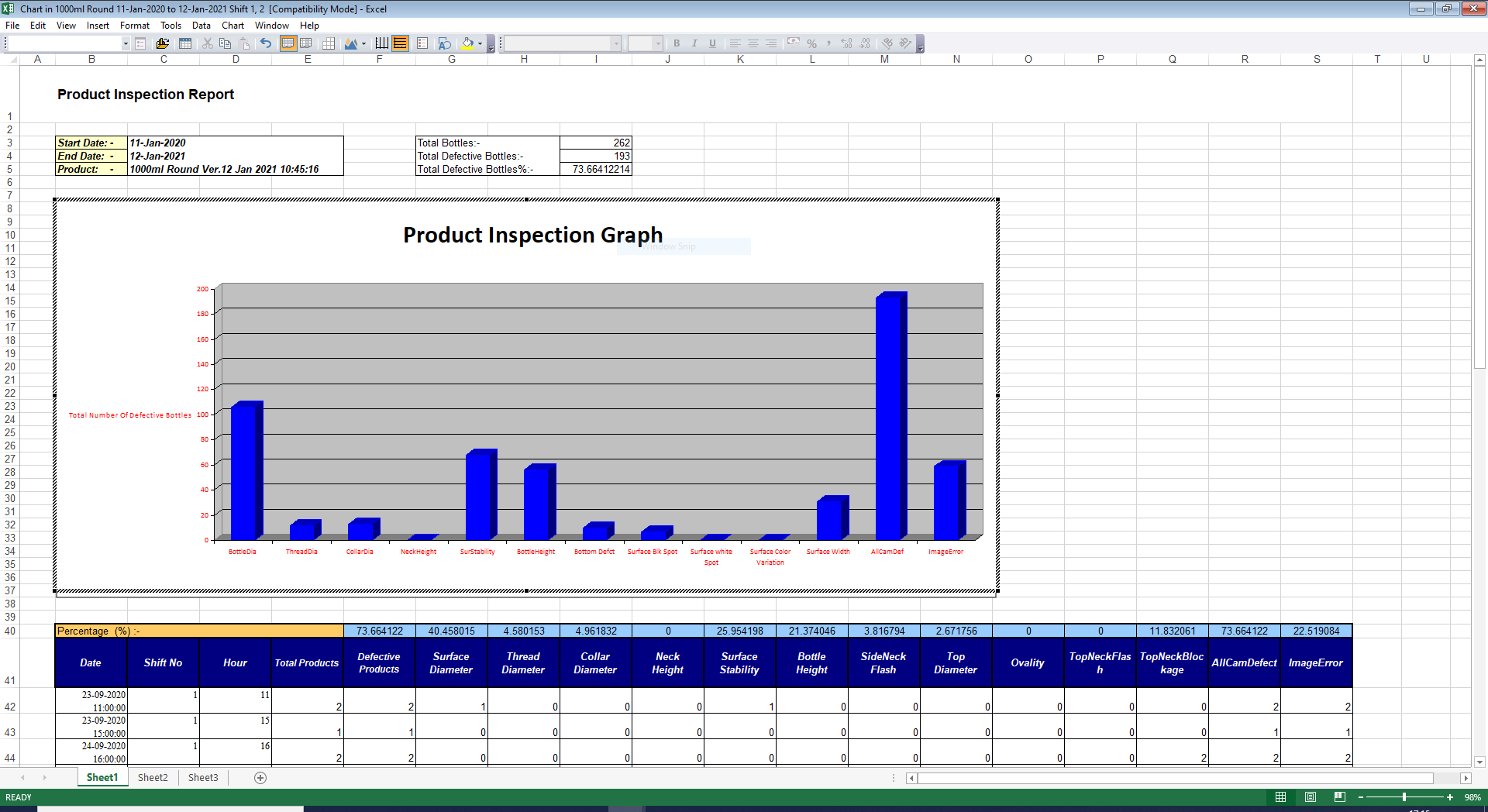
Reporting
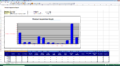
- The inspection screen displays graphically the last 100 detected defects to enable you to discover and fix potential problems in real time.
- All inspection data/results is stored in the database.
- Reports enable statistical process control of the induction sealing Process over time.
- User access command log report can be generated.
- Reports can be generated in both PDF and Excel formats.
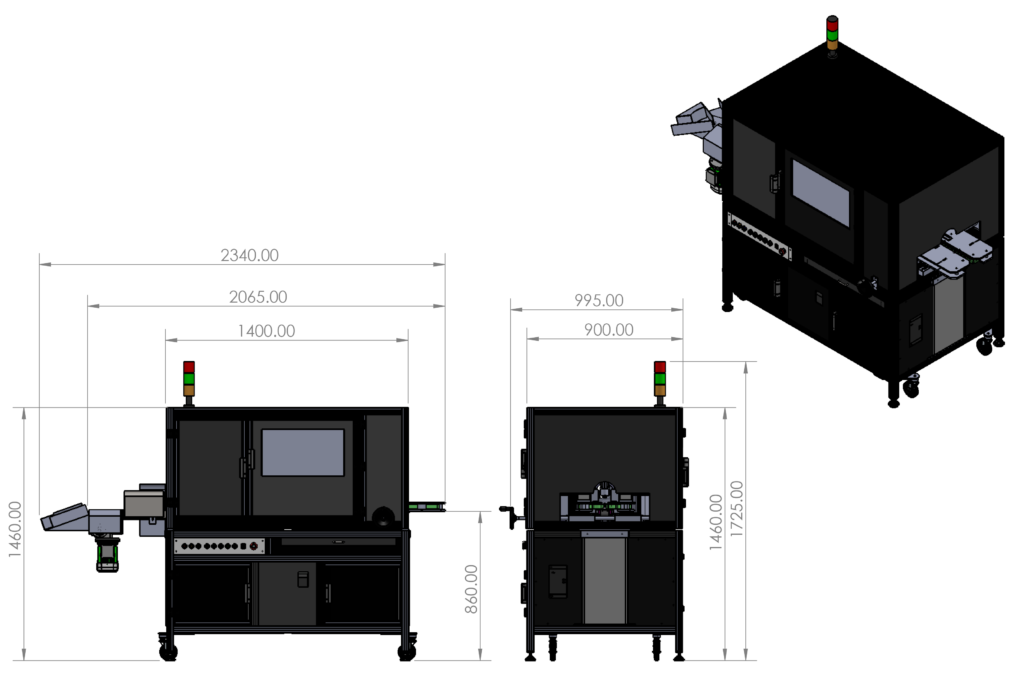
Machine Dimension
Software Screenshot